Confounded dose-response effects of treatment adherence: fitting Bayesian instrumental variable models using brms
Something that never ceases to amaze (depress) me, is how extremely common it is to see causal claims in RCTs, that are not part of the randomization. For instance, the relationship between treatment adherence and outcome, or between alliance and outcome, are often analyzed but seldom experimentally manipulated. This is basically observational research disguised as experimental, but without DAGs, instrumental variables, propensity scores, or any other technique used in observational research. Hence, the effects will almost surely be biased in an unknown direction. This is a post I’ve been planning to write for a long time, but put off since the theory and methods are so well covered in, for instance, epidemiology. However, when brms added support for multivariate formulas, I figured I could show how some of the techniques can be implemented in R, and their Bayesian counterparts using brms. So, in this post I do three things:
- Rant about how quickly clinical psychology researchers forget about causal inference, once they start analyzing non-randomized variables, such as the effect of treatment adherence (or “compliance”) on treatment outcome in a randomized controlled trial.
- Show that common regression methods either, makes absolutely no sense, or result in badly biased estimates of the causal dose-response effect, when a third variable influences both adherence and outcome, i.e. when the dose-response relationship is confounded. While instrumental variable methods may allow for unbiased estimates in these scenarios.
- Show how Bayesian intrumental variable regression can be easily fit using
brmsnew multivariate syntax. And also do a small simulation study to compare the results tolavaan,AER::ivregand biasedlm()models.
Motivating example
The effect of treatment adherence (psychotherapy dose-response effects)
Let’s assume we’ve performed an RCT and compared some psychotherapy condition to some control condition. Patients in these types of trials seldom complete all scheduled sessions, or adhere 100 % to the treatment. It’s rather common for researchers to try to see if there is some relationship between adherence and treatment outcome. Which is a valid question, if there is no added effect of actually “receiving” more of the treatment, then that should be a problem for your theory, or at least some of the components of that theory. However, what usually happens is that researchers somehow forget that the variable “adherence to therapy” is not experimentally manipulated. Unless we have randomized patients to receive 1, 2, …, 10 sessions, or to do 1, 2, …, 10, exposure exercises (and somehow ensured adherence was 100% in all conditions)–then we are now longer in RCT country!

Never stop to acknowledge you’re doing non-randomized comparisons!
A simple model of confounded adherence effects
A really simple scenario with one know (or unknown) confounder is shown in the DAG below.
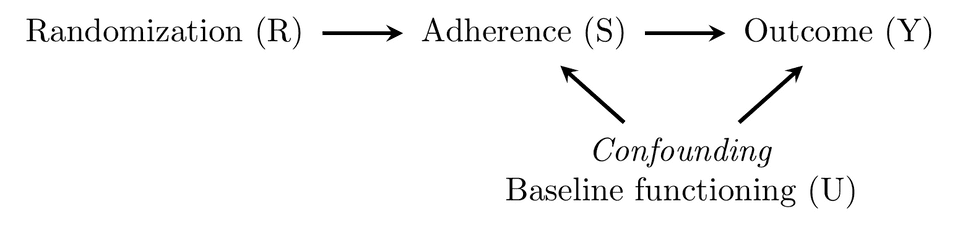
Each arrow represent causal influence of one variable on another. Y is the observed outcome, adherence is often measured by some proxy, like the number of sessions completed–that we assume is a good indicator of treatment “dose”, it could be any other proxy for treatment adherence. So, what the graph is saying, is that treatment allocation (randomization) influences the outcome (Y) via the amount of treatment received (adherence). Thus, unless something went horribly wrong, randomization should be strongly correlated with S, and only influence Y through the treatment received (S). By definition this makes randomization an instrumental variable. However, the problem is that the effect of S on Y is most likely confounded (U). For instance, baseline functioning might be related both to a better prognosis (better outcome) and treatment adherence. It is easy to come up with scenarios where the relationship could be either positive or negative. Higher baseline functioning could be related to a better prognosis, and to better treatment adherence, or, a patient could be too depressed to actually adhere to the treatment. It is also plausible that worse baseline functioning make you more motivated to adhere to your treatment. Whatever the relationship might be, it is clear that the causal effect of treatment adherence will be confounded.
We will deal with three types of scenarios where a variable confounds the estimate of treatment adherence:
- Observed confounder: is an observed variable that is adjusted for.
- Residual confounding: our known confounder is measured with error.
- Hidden confounding: is either unknown or unmeasured, and not adjusted for.
The data generating model
First, let’s remind ourselves what is commonly estimated in an RCT is the average treatment effect (ATE), or some related estimate,
Where is the would-be outcome for patient i after participating in the control condition, and the would be outcome for patient i after participating in the treatment condition. Obviously, we can never observe both and for the same patient. But thanks to randomization when can infer the ATE. We also assume that the individual treatment effect is the same for all individuals, given the same level of adherence.
The data generating model (DGM) is, thus, that adherence depends on , the confounder variable,
Where is just a function that centers and scales the confounding variable, so that the mean adherence is 7, and is just random variation that influence the relationship between and . Moreover, the observed measure of adherence could be a noisy measure of a patient’s actual adherence, , e.g. sessions completed,
To generate the outcome, we first simulate , and then calculate ,
So that is the causal effect of treatment adherence,
Lastly, the outcome is generated as,
Where is 1 for participants allocated to the treatment condition and 0 otherwise.
Different ways to answer the same question
Now, that we have a DGM, let’s generate some fake data and fit different models. We’ll start with a standard OLS regression, the first model would make sense in the absence of any hidden or residual confounding (extremely unlikely). The second model (“naïve OLS”) makes little sense, since it only analyze the treatment group, and as we’ll see, further confounds prognosis with the causal effect of treatment adherence. Sadly, this way of analyzing psychotherapy trials is really common… Anyway, lets generate some fake data. I’ll use a two-level random intercept-only model as a starting point, and assume that the confounder (baseline functioning), is measured without error.
OLS
First, we fit the two standard OLS models where we are adjusting for pretest scores.
Instrumental variables (IV)
Now, let’s try to use treatment assignment as an instrument to better uncover the causal effect of adherence. Two-stage least-squares (2SLS) is a common method to estimate an IV model. The point of this post is not to explain IV regression, it’s explained well in standard econometric textbooks (Wooldridge, 2010; Wooldridge, 2015). And in the context of psychotherapy research in the articles by Maracy and Dunn (2011), or Dunn and Bentall (2007). The very short explanation, is that 2SLS is basically to regress session on the instruments, then regress y on the predicted sessions from Step 1. This can be done using lm() to get point estimates, but the standard errors will be highly suspicious.
However, 2SLS is easier done with ivreg() from the package AER. The same structural model is fit using this code.
The model can also be thought of as a special case of a structural equation model, and fit with e.g. lavaan.
Lastly, using the same approach, a Bayesian IV model can be fit using brms (>2.0.0).
This allows extending the model a lot using brms highly flexible modelling syntax. Moreover, once you’ve exhausted brms capabilities you can get the stancode() and further customize the model yourself.
A small simulation study
Let’s also do a small simulation study, to better show the impact of observed confounding, residual confounding, and hidden confounding. We’ll use the OLS and IV models shown above. For each of the three scenarios, 5000 data sets were generated, with 50 patients per condition. The correlations between the variables are shown in the figure below, the actual values are arbitrary chosen, and the resulting bias, and its direction, is not meant to reflect real data, instead the values are meant to highlight the different types of confounding.
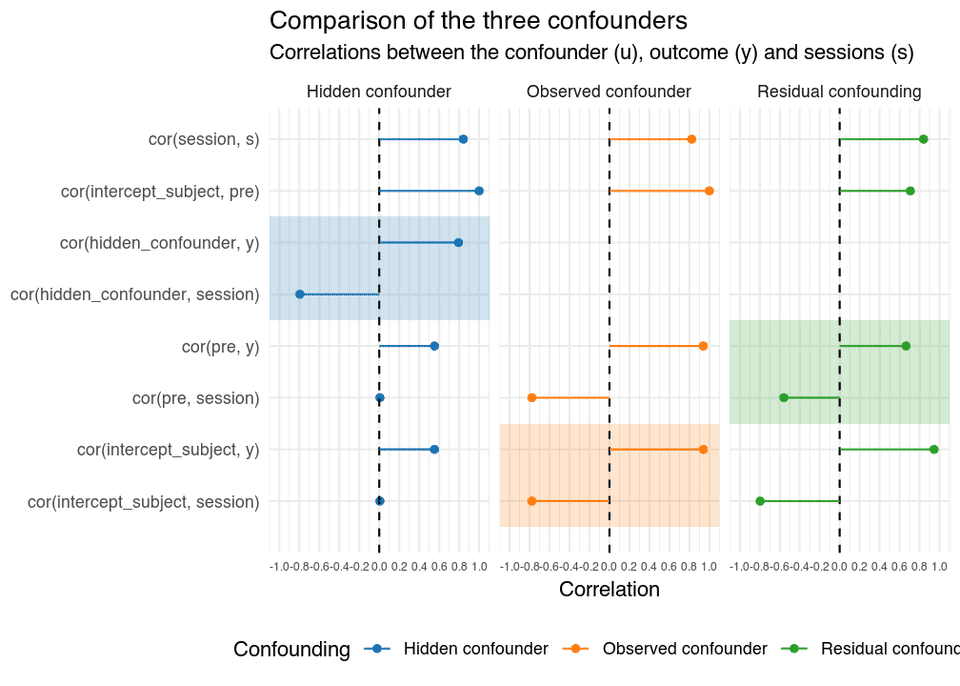
In the figure, s is the latent adherence variable, session the observed adherence, intercept_subject is the baseline confounder, that is measured with or without error by pre, and y is the outcome.
Relative bias
The relative bias of the estimated adherence effect is shown in the figure below.
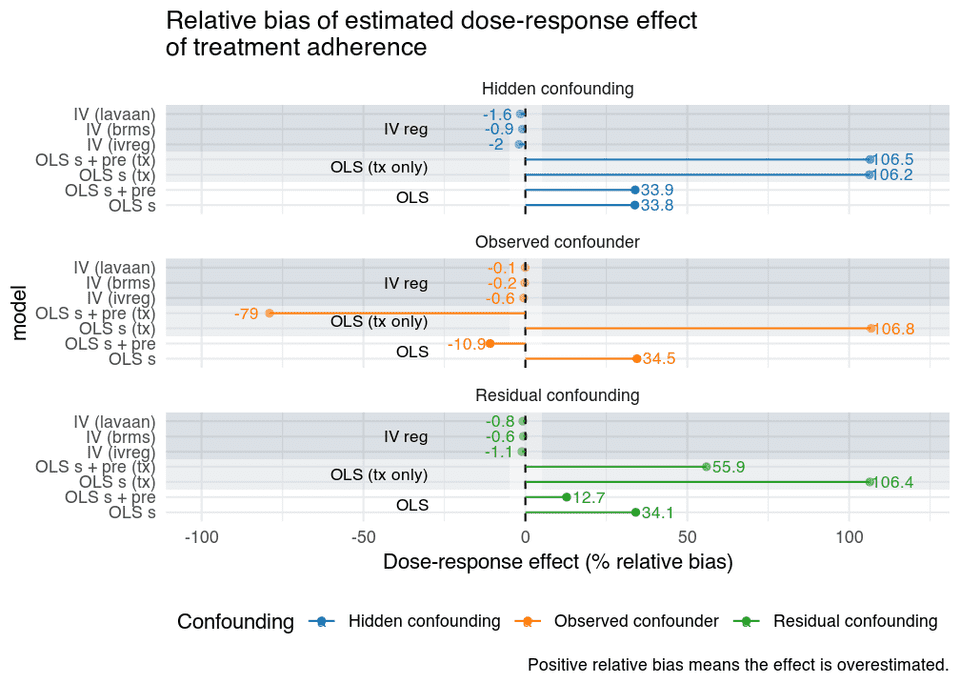
In the figure, we see for the OLS models, that when there’s:
- Hidden confounding:
- Adjusting for the baseline variable does not help, which is as expected, since it is not related to adherence.
- Observed confounder:
- The estimates are biased when the confounder is omitted.
- Adjusting for the confounder removes the bias. However, we do not recover the true causal effect, since session is a noisy proxy of adherence the effect is attenuated.
- Residual confounding:
- Controlling for the confounder reduces bias, but due to residual confounding estimates are still biased.
And the overall pattern is that:
- the naïve OLS is as bad idea, and is the most affected by bias in all scenarios. We use control groups for a reason, don’t ignore the control group!
- IV estimates are unbiased.
- However, large measurement errors in session would also attenuate the IV estimate.
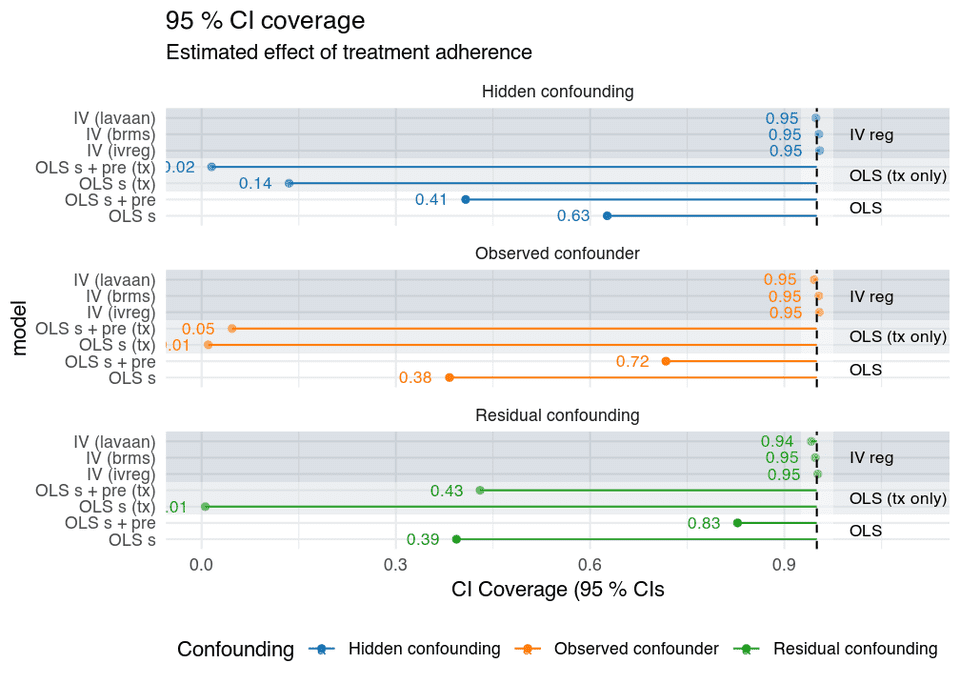
We see the same results if look at the coverage probabilities of the confidence intervals.
Coverage probabilities of the CIs
When less is more–collider bias
Let’s throw in another example of how things can go wrong, this time by adjusting for a collider. We extend the first DAG, and add another covariate therapeutic alliance. A researcher might think that by controlling for X (alliance), the dose-response relationship will be more clearly revealed. Or they might just do a multivariable regression and think “hey, both therapeutic alliance and adherence probably predict treatment outcome, let’s throw both in and see what p-value we get”. However, the problem is that alliance might neither be a predictor of outcome, nor a confounder of the adherence and outcome relationship, instead it could be a common effect of adherence and a hidden confounder (i.e., a collider). Obviously, this example is made up and alliance is just a placeholder–there’s so much going on during a psychological treatment that it would be easy to come up with other colliders. In the DAG bellow, conditioning on alliance opens up a path between adherence and the hidden confounder, that bias the relationship between adherence and outcome even more.
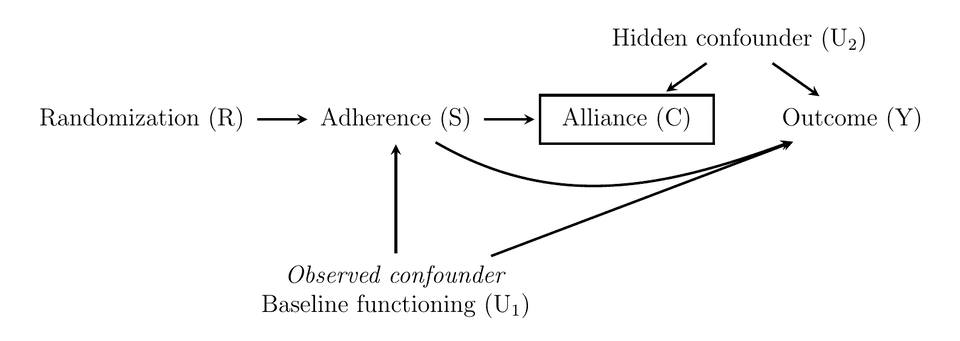
If we also simulate this model, and assume that is measured with error, and use the same IV regressions, but add an OLS model that also includes the collider we get these results.
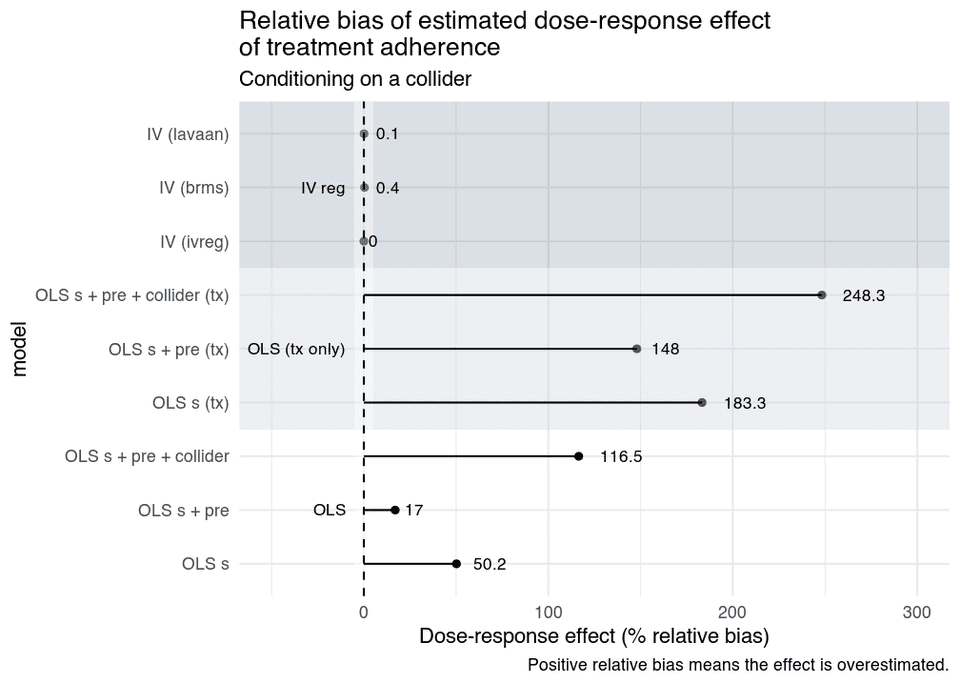
What we see is that:
- The OLS models that control for the baseline confounder is still biased due to residual confounding, and the naïve OLS is badly biased.
- By adjusting for the collider the bias increase, it would even have been better to not adjust for anything.
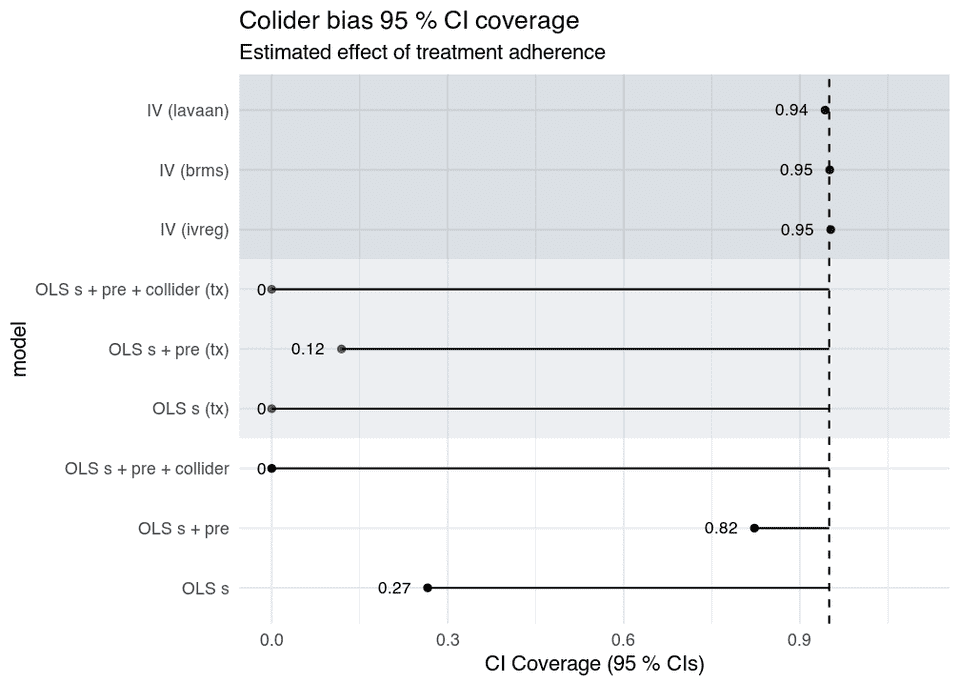
- The IV models are unbiased, and as you see below, their CIs have nominal coverage probabilities as well.
Summary
Lastly, to summarize this post:
- Don’t do naïve regressions on treatment adherence dose-response effects. It makes no sense even in the absence of confounding.
- Measurement error will bias your regression estimates even if you adjust for confounders.
- Confounders and colliders are everywhere, make your assumptions explicit. We don’t need more zombie studies claiming XYZ is related to treatment outcome.
- IV regression does not automagically solve all problems. But if you think hard about your data it can be a very useful tool in treatment studies.
brmsnew multivariate syntax make it extremely easy to fit Bayesian IV models, that can be extended to much more complicated scenarios than what I covered in this article.
References
- Dunn, G., & Bentall, R. (2007). Modelling treatment-effect heterogeneity in randomized controlled trials of complex interventions (psychological treatments). Statistics in Medicine, 26(26), 4719–4745. https://doi.org/10.1002/sim.2891
- Mohammad Maracy, & Graham Dunn. (2011). Estimating dose-response effects in psychological treatment trials: the role of instrumental variables. Statistical Methods in Medical Research, 20(3), 191–215. https://doi.org/10.1177/0962280208097243
- Wooldridge, J. M. (2010). Econometric analysis of cross section and panel data. MIT press.
- Wooldridge, J. M. (2015). Introductory econometrics: A modern approach. Nelson Education.
Code
Simulation functions
Setup and run the simulation.
Written by Kristoffer Magnusson, a researcher in clinical psychology. You should follow him on Bluesky or on Twitter.
Published February 01, 2018 (View on GitHub)
Buy Me A Coffee
A huge thanks to the 175 supporters who've bought me a 422 coffees!
Steffen bought ☕☕☕☕☕☕☕☕☕☕☕☕ (12) coffees
I love your visualizations. Some of the best out there!!!
Jason Rinaldo bought ☕☕☕☕☕☕☕☕☕☕ (10) coffees
I've been looking for applets that show this for YEARS, for demonstrations for classes. Thank you so much! Students do not need to tolarate my whiteboard scrawl now. I'm sure they'd appreciate you, too.l
Shawn Bergman bought ☕☕☕☕☕ (5) coffees
Thank you for putting this together! I am using these visuals and this information to teach my Advanced Quant class.
anthonystevendick@gmail.com bought ☕☕☕☕☕ (5) coffees
I've been using a lot of your ideas in a paper I'm writing and even borrowed some of your code (cited of course). But this site has been so helpful I think, in addition, I owe you a few coffees!
Chip Reichardt bought ☕☕☕☕☕ (5) coffees
Hi Krisoffer, these are great applets and I've examined many. I'm writing a chapter for the second edition of "Teaching statistics and quantitative methods in the 21st century" by Joe Rodgers (Routledge). My chapter is on the use of applets in teaching statistics. I could well be describing 5 of yours. Would you permit me to publish one or more screen shots of the output from one or more of your applets. I promise I will be saying very positive things about your applets. If you are inclined to respond, my email address if Chip.Reichardt@du.edu.
Someone bought ☕☕☕☕☕ (5) coffees
Someone bought ☕☕☕☕☕ (5) coffees
Nice work! Saw some of your other publications and they are also really intriguing. Thanks so much!
JDMM bought ☕☕☕☕☕ (5) coffees
You finally helped me understand correlation! Many, many thanks... 😄
@VicCazares bought ☕☕☕☕☕ (5) coffees
Good stuff! It's been so helpful for teaching a Psych Stats class. Cheers!
Dustin M. Burt bought ☕☕☕☕☕ (5) coffees
Excellent and informative visualizations!
Someone bought ☕☕☕☕☕ (5) coffees
@metzpsych bought ☕☕☕☕☕ (5) coffees
Always the clearest, loveliest simulations for complex concepts. Amazing resource for teaching intro stats!
Ryo bought ☕☕☕☕☕ (5) coffees
For a couple years now I've been wanting to create visualizations like these as a way to commit these foundational concepts to memory. But after finding your website I'm both relieved that I don't have to do that now and pissed off that I couldn't create anything half as beautiful and informative as you have done here. Wonderful job.
Diarmuid Harvey bought ☕☕☕☕☕ (5) coffees
You have an extremely useful site with very accessible content that I have been using to introduce colleagues and students to some of the core concepts of statistics. Keep up the good work, and thanks!
Michael Hansen bought ☕☕☕☕☕ (5) coffees
Keep up the good work!
Michael Villanueva bought ☕☕☕☕☕ (5) coffees
I wish I could learn more from you about stats and math -- you use language in places that I do not understand. Cohen's D visualizations opened my understanding. Thank you
Someone bought ☕☕☕☕☕ (5) coffees
Thank you, Kristoffer
Pål from Norway bought ☕☕☕☕☕ (5) coffees
Great webpage, I use it to illustrate several issues when I have a lecture in research methods. Thanks, it is really helpful for the students:)
@MAgrochao bought ☕☕☕☕☕ (5) coffees
Joseph Bulbulia bought ☕☕☕☕☕ (5) coffees
Hard to overstate the importance of this work Kristoffer. Grateful for all you are doing.
@TDmyersMT bought ☕☕☕☕☕ (5) coffees
Some really useful simulations, great teaching resources.
@lakens bought ☕☕☕☕☕ (5) coffees
Thanks for fixing the bug yesterday!
@LinneaGandhi bought ☕☕☕☕☕ (5) coffees
This is awesome! Thank you for creating these. Definitely using for my students, and me! :-)
@ICH8412 bought ☕☕☕☕☕ (5) coffees
very useful for my students I guess
@KelvinEJones bought ☕☕☕☕☕ (5) coffees
Preparing my Master's student for final oral exam and stumbled on your site. We are discussing in lab meeting today. Coffee for everyone.
Someone bought ☕☕☕☕☕ (5) coffees
What a great site
@Daniel_Brad4d bought ☕☕☕☕☕ (5) coffees
Wonderful work!
David Loschelder bought ☕☕☕☕☕ (5) coffees
Terrific work. So very helpful. Thank you very much.
@neilmeigh bought ☕☕☕☕☕ (5) coffees
I am so grateful for your page and can't thank you enough!
@giladfeldman bought ☕☕☕☕☕ (5) coffees
Wonderful work, I use it every semester and it really helps the students (and me) understand things better. Keep going strong.
Dean Norris bought ☕☕☕☕☕ (5) coffees
Sal bought ☕☕☕☕☕ (5) coffees
Really super useful, especially for teaching. Thanks for this!
dde@paxis.org bought ☕☕☕☕☕ (5) coffees
Very helpful to helping teach teachers about the effects of the Good Behavior Game
@akreutzer82 bought ☕☕☕☕☕ (5) coffees
Amazing visualizations! Thank you!
@rdh_CLE bought ☕☕☕☕☕ (5) coffees
So good!
tchipman1@gsu.edu bought ☕☕☕ (3) coffees
Hey, your stuff is cool - thanks for the visual
Hugo Quené bought ☕☕☕ (3) coffees
Hi Kristoffer, Some time ago I've come up with a similar illustration about CIs as you have produced, and I'm now also referring to your work:<br>https://hugoquene.github.io/QMS-EN/ch-testing.html#sec:t-confidenceinterval-mean<br>With kind regards, Hugo Quené<br>(Utrecht University, Netherlands)
Tor bought ☕☕☕ (3) coffees
Thanks so much for helping me understand these methods!
Amanda Sharples bought ☕☕☕ (3) coffees
Soyol bought ☕☕☕ (3) coffees
Someone bought ☕☕☕ (3) coffees
Kenneth Nilsson bought ☕☕☕ (3) coffees
Keep up the splendid work!
@jeremywilmer bought ☕☕☕ (3) coffees
Love this website; use it all the time in my teaching and research.
Someone bought ☕☕☕ (3) coffees
Powerlmm was really helpful, and I appreciate your time in putting such an amazing resource together!
DR AMANDA C DE C WILLIAMS bought ☕☕☕ (3) coffees
This is very helpful, for my work and for teaching and supervising
Georgios Halkias bought ☕☕☕ (3) coffees
Regina bought ☕☕☕ (3) coffees
Love your visualizations!
Susan Evans bought ☕☕☕ (3) coffees
Thanks. I really love the simplicity of your sliders. Thanks!!
@MichaMarie8 bought ☕☕☕ (3) coffees
Thanks for making this Interpreting Correlations: Interactive Visualizations site - it's definitely a great help for this psych student! 😃
Zakaria Giunashvili, from Georgia bought ☕☕☕ (3) coffees
brilliant simulations that can be effectively used in training
Someone bought ☕☕☕ (3) coffees
@PhysioSven bought ☕☕☕ (3) coffees
Amazing illustrations, there is not enough coffee in the world for enthusiasts like you! Thanks!
Cheryl@CurtinUniAus bought ☕☕☕ (3) coffees
🌟What a great contribution - thanks Kristoffer!
vanessa moran bought ☕☕☕ (3) coffees
Wow - your website is fantastic, thank you for making it.
Someone bought ☕☕☕ (3) coffees
mikhail.saltychev@gmail.com bought ☕☕☕ (3) coffees
Thank you Kristoffer This is a nice site, which I have been used for a while. Best Prof. Mikhail Saltychev (Turku University, Finland)
Someone bought ☕☕☕ (3) coffees
Ruslan Klymentiev bought ☕☕☕ (3) coffees
@lkizbok bought ☕☕☕ (3) coffees
Keep up the nice work, thank you!
@TELLlab bought ☕☕☕ (3) coffees
Thanks - this will help me to teach tomorrow!
SCCT/Psychology bought ☕☕☕ (3) coffees
Keep the visualizations coming!
@elena_bolt bought ☕☕☕ (3) coffees
Thank you so much for your work, Kristoffer. I use your visualizations to explain concepts to my tutoring students and they are a huge help.
A random user bought ☕☕☕ (3) coffees
Thank you for making such useful and pretty tools. It not only helped me understand more about power, effect size, etc, but also made my quanti-method class more engaging and interesting. Thank you and wish you a great 2021!
@hertzpodcast bought ☕☕☕ (3) coffees
We've mentioned your work a few times on our podcast and we recently sent a poster to a listener as prize so we wanted to buy you a few coffees. Thanks for the great work that you do!Dan Quintana and James Heathers - Co-hosts of Everything Hertz
Cameron Proctor bought ☕☕☕ (3) coffees
Used your vizualization in class today. Thanks!
eshulman@brocku.ca bought ☕☕☕ (3) coffees
My students love these visualizations and so do I! Thanks for helping me make stats more intuitive.
Someone bought ☕☕☕ (3) coffees
Adrian Helgå Vestøl bought ☕☕☕ (3) coffees
@misteryosupjoo bought ☕☕☕ (3) coffees
For a high school teacher of psychology, I would be lost without your visualizations. The ability to interact and manipulate allows students to get it in a very sticky manner. Thank you!!!
Chi bought ☕☕☕ (3) coffees
You Cohen's d post really helped me explaining the interpretation to people who don't know stats! Thank you!
Someone bought ☕☕☕ (3) coffees
You doing useful work !! thanks !!
@ArtisanalANN bought ☕☕☕ (3) coffees
Enjoy.
@jsholtes bought ☕☕☕ (3) coffees
Teaching stats to civil engineer undergrads (first time teaching for me, first time for most of them too) and grasping for some good explanations of hypothesis testing, power, and CI's. Love these interactive graphics!
@notawful bought ☕☕☕ (3) coffees
Thank you for using your stats and programming gifts in such a useful, generous manner. -Jess
Mateu Servera bought ☕☕☕ (3) coffees
A job that must have cost far more coffees than we can afford you ;-). Thank you.
@cdrawn bought ☕☕☕ (3) coffees
Thank you! Such a great resource for teaching these concepts, especially CI, Power, correlation.
Julia bought ☕☕☕ (3) coffees
Fantastic work with the visualizations!
@felixthoemmes bought ☕☕☕ (3) coffees
@dalejbarr bought ☕☕☕ (3) coffees
Your work is amazing! I use your visualizations often in my teaching. Thank you.
@PsychoMouse bought ☕☕☕ (3) coffees
Excellent! Well done! SOOOO Useful!😊 🐭
Someone bought ☕☕ (2) coffees
Thanks, your work is great!!
Dan Sanes bought ☕☕ (2) coffees
this is a superb, intuitive teaching tool!
@whlevine bought ☕☕ (2) coffees
Thank you so much for these amazing visualizations. They're a great teaching tool and the allow me to show students things that it would take me weeks or months to program myself.
Someone bought ☕☕ (2) coffees
@notawful bought ☕☕ (2) coffees
Thank you for sharing your visualization skills with the rest of us! I use them frequently when teaching intro stats.
Someone bought ☕ (1) coffee
You are awesome
Thom Marchbank bought ☕ (1) coffee
Your visualisations are so useful! Thank you so much for your work.
georgina g. bought ☕ (1) coffee
thanks for helping me in my psych degree!
Someone bought ☕ (1) coffee
Thank You for this work.
Kosaku Noba bought ☕ (1) coffee
Nice visualization, I bought a cup of coffee.
Someone bought ☕ (1) coffee
Thomas bought ☕ (1) coffee
Great. Use it for teaching in psychology.
Someone bought ☕ (1) coffee
It is the best statistics visualization so far!
Ergun Pascu bought ☕ (1) coffee
AMAZING Tool!!! Thank You!
Ann Calhoun-Sauls bought ☕ (1) coffee
This has been a wonderful resource for my statistics and research methods classes. I also occassionally use it for other courses such as Theories of Personality and Social Psychology
David Britt bought ☕ (1) coffee
nicely reasoned
Mike bought ☕ (1) coffee
I appreciate your making this site available. Statistics are not in my wheelhouse, but the ability to display my data more meaningfully in my statistics class is both educational and visually appealing. Thank you!
Jayne T Jacobs bought ☕ (1) coffee
Andrew J O'Neill bought ☕ (1) coffee
Thanks for helping understand stuff!
Someone bought ☕ (1) coffee
Someone bought ☕ (1) coffee
Shawn Hemelstrand bought ☕ (1) coffee
Thank you for this great visual. I use it all the time to demonstrate Cohen's d and why mean differences affect it's approximation.
Adele Fowler-Davis bought ☕ (1) coffee
Thank you so much for your excellent post on longitudinal models. Keep up the good work!
Stewart bought ☕ (1) coffee
This tool is awesome!
Someone bought ☕ (1) coffee
Aidan Nelson bought ☕ (1) coffee
Such an awesome page, Thank you
Someone bought ☕ (1) coffee
Ellen Kearns bought ☕ (1) coffee
Dr Nazam Hussain bought ☕ (1) coffee
Someone bought ☕ (1) coffee
Eva bought ☕ (1) coffee
I've been learning about power analysis and effect sizes (trying to decide on effect sizes for my planned study to calculate sample size) and your Cohen's d interactive tool is incredibly useful for understanding the implications of different effect sizes!
Someone bought ☕ (1) coffee
Someone bought ☕ (1) coffee
Thanks a lot!
Someone bought ☕ (1) coffee
Reena Murmu Nielsen bought ☕ (1) coffee
Tony Andrea bought ☕ (1) coffee
Thanks mate
Tzao bought ☕ (1) coffee
Thank you, this really helps as I am a stats idiot :)
Melanie Pflaum bought ☕ (1) coffee
Sacha Elms bought ☕ (1) coffee
Yihan Xu bought ☕ (1) coffee
Really appreciate your good work!
@stevenleung bought ☕ (1) coffee
Your visualizations really help me understand the math.
Junhan Chen bought ☕ (1) coffee
Someone bought ☕ (1) coffee
Someone bought ☕ (1) coffee
Michael Hansen bought ☕ (1) coffee
ALEXANDER VIETHEER bought ☕ (1) coffee
mather bought ☕ (1) coffee
Someone bought ☕ (1) coffee
Bastian Jaeger bought ☕ (1) coffee
Thanks for making the poster designs OA, I just hung two in my office and they look great!
@ValerioVillani bought ☕ (1) coffee
Thanks for your work.
Someone bought ☕ (1) coffee
Great work!
@YashvinSeetahul bought ☕ (1) coffee
Someone bought ☕ (1) coffee
Angela bought ☕ (1) coffee
Thank you for building such excellent ways to convey difficult topics to students!
@inthelabagain bought ☕ (1) coffee
Really wonderful visuals, and such a fantastic and effective teaching tool. So many thanks!
Someone bought ☕ (1) coffee
Someone bought ☕ (1) coffee
Yashashree Panda bought ☕ (1) coffee
I really like your work.
Ben bought ☕ (1) coffee
You're awesome. I have students in my intro stats class say, "I get it now," after using your tool. Thanks for making my job easier.
Gabriel Recchia bought ☕ (1) coffee
Incredibly useful tool!
Shiseida Sade Kelly Aponte bought ☕ (1) coffee
Thanks for the assistance for RSCH 8210.
@Benedikt_Hell bought ☕ (1) coffee
Great tools! Thank you very much!
Amalia Alvarez bought ☕ (1) coffee
@noelnguyen16 bought ☕ (1) coffee
Hi Kristoffer, many thanks for making all this great stuff available to the community!
Eran Barzilai bought ☕ (1) coffee
These visualizations are awesome! thank you for creating it
Someone bought ☕ (1) coffee
Chris SG bought ☕ (1) coffee
Very nice.
Gray Church bought ☕ (1) coffee
Thank you for the visualizations. They are fun and informative.
Qamar bought ☕ (1) coffee
Tanya McGhee bought ☕ (1) coffee
@schultemi bought ☕ (1) coffee
Neilo bought ☕ (1) coffee
Really helpful visualisations, thanks!
Someone bought ☕ (1) coffee
This is amazing stuff. Very slick.
Someone bought ☕ (1) coffee
Sarko bought ☕ (1) coffee
Thanks so much for creating this! Really helpful for being able to explain effect size to a clinician I'm doing an analysis for.
@DominikaSlus bought ☕ (1) coffee
Thank you! This page is super useful. I'll spread the word.
Someone bought ☕ (1) coffee
Melinda Rice bought ☕ (1) coffee
Thank you so much for creating these tools! As we face the challenge of teaching statistical concepts online, this is an invaluable resource.
@tmoldwin bought ☕ (1) coffee
Fantastic resource. I think you would be well served to have one page indexing all your visualizations, that would make it more accessible for sharing as a common resource.
Someone bought ☕ (1) coffee
Fantastic Visualizations! Amazing way to to demonstrate how n/power/beta/alpha/effect size are all interrelated - especially for visual learners! Thank you for creating this?
@jackferd bought ☕ (1) coffee
Incredible visualizations and the best power analysis software on R.
Cameron Proctor bought ☕ (1) coffee
Great website!
Someone bought ☕ (1) coffee
Hanah Chapman bought ☕ (1) coffee
Thank you for this work!!
Someone bought ☕ (1) coffee
Jayme bought ☕ (1) coffee
Nice explanation and visual guide of Cohen's d
Bart Comly Boyce bought ☕ (1) coffee
thank you
Dr. Mitchell Earleywine bought ☕ (1) coffee
This site is superb!
Florent bought ☕ (1) coffee
Zampeta bought ☕ (1) coffee
thank you for sharing your work.
Mila bought ☕ (1) coffee
Thank you for the website, made me smile AND smarter :O enjoy your coffee! :)
Deb bought ☕ (1) coffee
Struggling with statistics and your interactive diagram made me smile to see that someone cares enough about us strugglers to make a visual to help us out!😍
Someone bought ☕ (1) coffee
@exerpsysing bought ☕ (1) coffee
Much thanks! Visualizations are key to my learning style!
Someone bought ☕ (1) coffee
Sponsors
You can sponsor my open source work using GitHub Sponsors and have your name shown here.
Backers ✨❤️
Questions & Comments
Please use GitHub Discussions for any questions related to this post, or open an issue on GitHub if you've found a bug or wan't to make a feature request.
Archived Comments (2)
Great post dear. It definitely has increased my knowledge on R Programming. Please keep sharing similar write ups of yours. You can check this too for R Programming tutorial as i have recorded this recently on R Programming. and i'm sure it will be helpful to you.https://www.youtube.com/watch?v=rgF...
”Rant about how quickly clinical psychology researchers forget about casual inference"
Casual inference?